Loading...
Loading...
| Jan 01, 1970 | Daily Report |
| RISING BHARAT | News Count (102624) | |
20521. Over 550 scientists returned to India to pursue research in biotechnology: Centre
- Over 550 Indian biotechnologists have returned to their homeland to pursue careers in research here under the prestigious Ramalingaswami Re-entry Fellowship, the government said
- several more scientists abroad were keen to return to benefit from the research opportunities and careers in the country.
- The Ramalingaswami Re-entry Fellowship is a prestigious scheme of the Department of Biotechnology, which was launched in 2006-07 with the aim to bring back the Indian scientists working abroad.
Click here to find 1469 similar incidents of Bharat Economic Growth -> Bharat Economic Growth
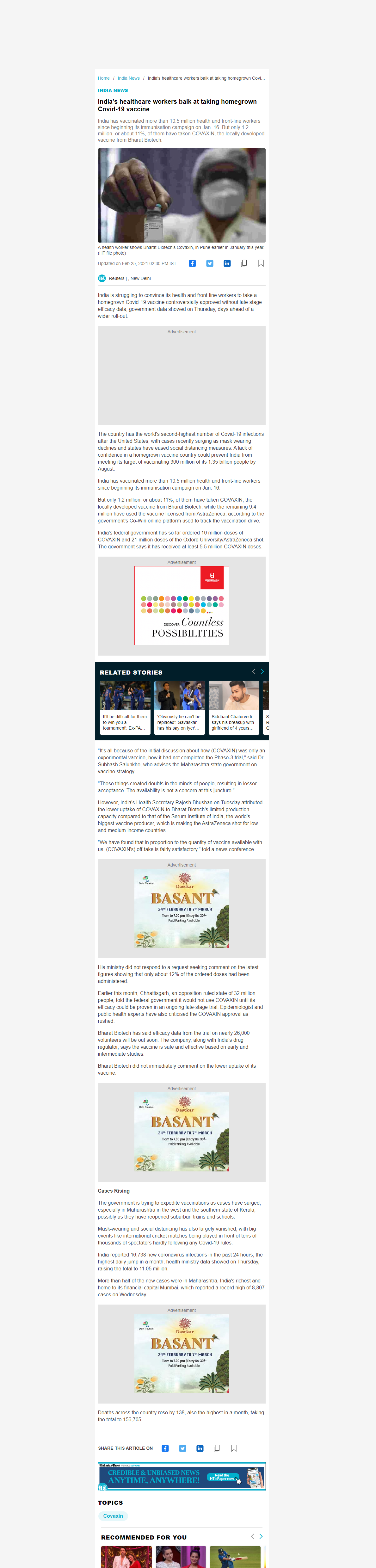
20522. India's healthcare workers balk at taking homegrown Covid-19 vaccine
- India has vaccinated more than 10.5 million health and front-line workers since beginning its immunisation campaign on Jan. 16. But only 1.2 million, or about 11%, of them have taken COVAXIN, the locally developed vaccine from Bharat Biotech.
- The country has the world's second-highest number of Covid-19 infections after the United States, with cases recently surging as mask wearing declines and states have eased social distancing measures. A lack of confidence in a homegrown vaccine country could prevent India from meeting its target of vaccinating 300 million of its 1.35 billion people by August.
- India's federal government has so far ordered 10 million doses of COVAXIN and 21 million doses of the Oxford University/AstraZeneca shot. The government says it has received at least 5.5 million COVAXIN doses.
20523. India's drugs regulator rejects Bharat Biotech's request for COVID-19 vaccine trials on children
- The drugs regulator's rejection came after hearing an application by Hyderabad-based pharma company on Wednesday sought approval for Phase 3 clinical trials of Covaxin on children between 5-18 years of age.
- "The efficacy data is top priority and they will also have to produce that data that has been asked for by the experts before more permissions are granted," local daily Hindustan Times quoted drugs controller officials as saying.
- Covaxin is already approved for use in India for adults in a clinical trial mode based on the safety and immunogenicity data the company submitted.
20524. Guava export rises to USD 2.09 million in Apr-Jan
- Export of guavas has risen to USD 2.09 million in April-January 2021-22 against USD 0.58 million in April-January 2013-14, the commerce ministry said
- The export of curd (yogurt) and paneer (Indian cottage cheese) has also increased from USD 10 million in April-January 2013-14 to USD 30 million in April-January 2021-22.
- Major exporting destinations for dairy products are the UAE , Bangladesh, the US, Bhutan, Singapore, Saudi Arabia, Malaysia, Qatar, Oman and Indonesia.
Click here to find 862 similar incidents of Bharat Economic Growth -> Increasing Exports
20525. African Union backs India's call to waive IP rights on Covid-19 drugs
- South Africa and India, which both manufacture drugs and vaccines, made the proposal at the WTO last year, arguing that intellectual property rules were hindering the urgent scale-up of vaccine production and provision of medical products to some patients.
- The African Union is backing calls for drugmakers to waive some intellectual property rights on Covid-19 medicines and vaccines to speed up their rollout to poor countries,
- They have faced opposition from some developed nations, but the backing of the African Union (AU) may give renewed impetus for the push to relax IP rules. John Nkengasong, director of the Africa Centers for Disease Control and Prevention, told a news conference that IP transfer was a "win-win for everybody" that would address the huge inequalities in global public health.
20526. Nagaland now officially has a rail tunnel
- Nagaland state can now officially claim to have an artificial tunnel in its turf. The Dhansiri-Zubza rail line witnessed the first tunnel breakthrough on June 7, roughly three years after the foundation stone for the project was laid in August 2016 by the then Union Railway Minister.
- The tunnel measured 80m in length and is located on the southern outskirts of Chumoükedima village some 20 minutes drive from NH 29.
- It also includes 19 major bridges, the longest of which, sited near Sirhima village, will span 700m.
Click here to find 759 similar incidents of North East India -> Infrastructure Development
20527. WHO ने आपातकालीन उपयोग के लिए चीन की साइनोफर्म Covid19 वैक्सीन को किया सूचीबद्ध
- विश्व स्वास्थ्य संगठन (WHO) ने सभी देशों में आपातकालीन उपयोग के लिए साइनोफर्म COVID19 वैक्सीन को सूचीबद्ध कर दिया है. जो संभावित COVAX रोल-आउट के लिए एक शर्त है.
- डब्ल्यूएचओ के महानिदेशक टेड्रोस अधनोम ग्रेबेसियस ने कहा कि 'डब्ल्यूएचओ ने बीजिंग के COVID-19 वैक्सीन पर हस्ताक्षर करने के लिए आपातकालीन उपयोग सूची जारी कर दी है, जिससे यह सुरक्षा क्षमता और गुणवत्ता के लिए WHO सत्यापन प्राप्त करने वाला छठा टीका बन गया.
- डब्ल्यूएचओ के तकनीकी सलाहकार समूह ने एक चीनी साइनोफर्म COVID19 वैक्सीन को संयुक्त राष्ट्र समर्थित COVAX कार्यक्रम में शामिल किया है.
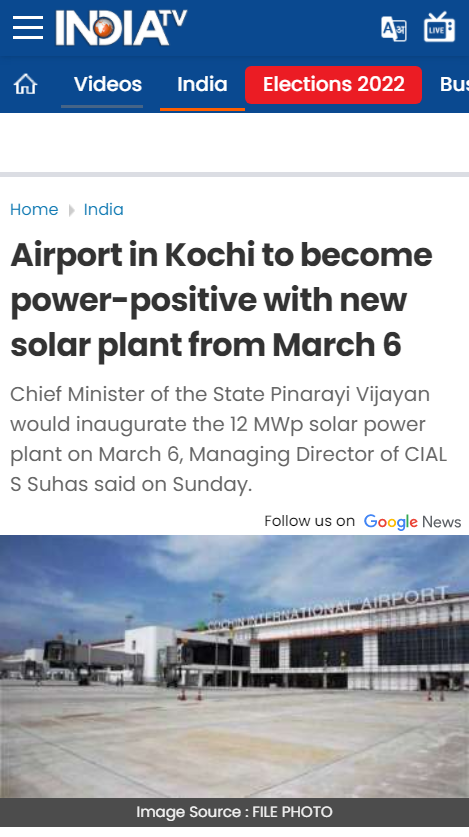
20528. Airport in Kochi to become power-positive with new solar plant from March 6
- Cochin International Airport, world's 1st airport powered by solar energy, to become power positive
- Power plant boasts of 12 megawatt capacity on a 35-acre land
- the carbon footprint would be reduced by 28,000 metric tonnes per year. The airport became the first in the world in 2015 to be fully powered by solar energy.
20529. भारत को सब्सिडी के साथ मिलेंगे कोरोना के 19 से 25 करोड़ वैक्सीन- GAVI
- ग्लोबल एलायंस फॉर वैक्सीन एंड इम्यूनिशन (गावी) की ओर से बयान जारी कर कहा गया है कि भारत को सब्सिडी के साथ 19 से 25 करोड़ कोरोना के टीके दिए जाएंगे.
- गावी कम और मध्यम आय वाले देशों को कोरोना का टीका दिलवाने के लिए वैश्विक स्तर पर प्रयास में जुटा हुआ है.
- गावी की ओर से बयान जारी कर कहा गया है कि भारत को तात्कालिक तकनीकी सहायता और कूलिंग चेन उपकरण के लिए 3 करोड़ डॉलर की वित्तीय मदद भी दी जाएगी.
20530. Zydus Cadila इस महीने मांग सकती है अपनी कोरोना वैक्सीन के लिए अनुमति
- मेडिसिन बनाने वाली अहमदाबाद की प्रमुख कंपनी जायडस कैडिला इस महीने अपनी कोरोना वैक्सीन के लिए अनुमति मांग सकती है. कंपनी को कोविड वैक्सीन से जुड़ा डेटा मिल गया है. अब इस टीके के लिए इमरजेंसी यूज ऑथराइजेशन का आवेदन कर सकती है.
- जायडस कैडिला के मैनेजिंग डायरेक्टर (MD) शर्विल पटेल ने कहा, "हम इस महीने रेगुलेटर को ट्रायल का डेटा देकर अनुमति मांग सकते हैं. हमें इसी महीने इमरजेंसी यूज ऑथराइजेशन की अनुमति मिल सकती है."
- कंपनी ने प्लासमिड DNA वैक्सीन के लिए तीसरे फेज का क्लिनिकल ट्रायल फरवरी में शुरू किया था. इसमें 28,000 पार्टिसिपेंट को शामिल किया गया था.
- अभी तक देश में वैक्सीन की दो डोज लेने की जरूरत होती है, लेकिन जायडस कैडिला के वैक्सीन की तीन डोज की टेस्टिंग की गई.
- ये तीनों डोज एक महीने के अंतराल पर लेनी होगी. इसके अलावा कंपनी दो डोज वाली वैक्सीन ZyCoV-D का भी ट्रायल कर रही है. ये ट्रायल भी मई में पूरा होने की उम्मीद है.