Loading...
Loading...
| Jan 01, 1970 | Daily Report |
| RISING BHARAT | News Count (102626) | |
20481. This New York-based cleantech startup aims to create premium drinking water for all
Innovation and startups, ISRO/Defense/Water infrastructure/EV and other sectors
->
Startups in Bharat
English
India
Your Story
|
- New York-based cleantech startup Kara Water provides clean drinking water solutions to address the growing demand for premium water enriched with minerals free from harmful contaminants usually found in tap water.
- The New York-headquartered startup — founded in 2017 by Michael Di Giovanna and Cody — provides clean drinking water solutions to address the growing demand for premium water enriched with minerals free from harmful contaminants usually found in tap water.
- The startup sells Kara Pure for Rs 135,000 and offers EMI services that allow customers to pay in monthly installments.
20482. 165 lakh COVID vaccine doses procured so far at cost of Rs 350.25 crore: MoS Health Ashwini Choubey
India Fights Corona: Vaccination Drive and Initiatives
English
India
The New Indian Express
|
- A total of 165 lakh doses of COVID-19 vaccines - Covishield and Covaxin - have been procured so far at a cost of Rs 350.25 crore, Minister of State for Health Ashwini Choubey told Lok Sabha .
- only these two vaccines have been procured for COVID-19 vaccination drive in the country. A total of 165 lakh doses of COVID-19 vaccine have been procured so far, of which 110 lakh doses are of Covishield and 55 lakh doses are of Covaxin vaccine. The total cost of procurement of above mentioned doses of vaccines is Rs 350.25 crore," he said.
- The existing infrastructure under Universal Immunization Programme is being used and simultaneously strengthened for deployment of vaccines, including storage and transportation.
20483. 14 EU Parliament Members Back India And South Africa's COVID-19 Vaccine Patent Proposal
India Fights Corona: Vaccination Drive and Initiatives
English
India
Republic
|
- In October 2020, India and South Africa in a joint statement had submitted a communication at The Agreement on Trade-Related Aspects of Intellectual Property (TRIPS) that was reportedly titled 'Waiver from certain provisions of the TRIPS agreement for the prevention, containment, and treatment of COVID-19'. The nations jointly requested a waiver to be granted to WTO members so that they do not have to implement, apply or enforce certain obligations related to the COVID-19 products and technologies.
- The letter was sent by European Parliament members including Andrea Cozzolino, Maria Arena, Eva Kaili, Alex Agius Saliba, Boguslaw Liberadzki, Tiemo Woelken, Milan Brglez, Patrizia Toia, Irene Tinagli, Pierfrancesco Majorino, Pina Picierno, Giuliano Pisapia, Massimiliano Smeriglio, Franco Roberti.
- In October 2020, India and South Africa in a joint statement had submitted a communication at The Agreement on Trade-Related Aspects of Intellectual Property (TRIPS) that was reportedly titled 'Waiver from certain provisions of the TRIPS agreement for the prevention, containment, and treatment of COVID-19.
20484. Plan to increase Sputnik V vaccine production up to 850 million doses per year in India: Russian envoy
India Fights Corona: Vaccination Drive and Initiatives
English
Russian Federation
ANI
|
- Expressing his happiness at the delivery of the first batch of Sputnik V vaccine to India, Russian envoy Nikolay Kudashev on Saturday said that the local production is about to start soon and is planned to be gradually increased up to 850 million doses per year.
- Earlier today, a flight carrying the first lot of Russian vaccines landed in Hyderabad, Telangana.
- .
20485. 21 days into Covid vaccination drive, India inoculates 5 million beneficiaries
India Fights Corona: Vaccination Drive and Initiatives
English
India
Mint
|
- 50 lakh people have been vaccinated in the 21 days since inoculation drive began in India on 16 January, the union health ministry said .
- A total of 52,90,474 healthcare workers have been vaccinated until 6 pm on 5 February in 1,04,781 sessions.
- As many as 3,31,029 beneficiaries were vaccinated on Friday, according to the health ministry. Agnani further said that no person was hospitalised after vaccination in the last 24 hours. "Total hospitalisation till date is 27, no hospitalisation in past 24 hours. Deaths so far are 22.
20486. India fastest to cross 5 million-mark on Covid vaccination, says health ministry
India Fights Corona: Vaccination Drive and Initiatives
English
India
Hindustan Times
|
- "As on February 5, 2021, 6pm, a total of 52,90,474 beneficiaries have been vaccinated for Covid-19 and for which a total of 1,04,781 sessions have been held," said Manohar Agnani, additional secretary at the health ministry.
- India is the fastest to inoculate more than 5 million beneficiaries against the deadly virus. In just 21 days, India vaccinated over 5.2 million beneficiaries, while the US took 24 days, the UK 43 days and Israel took 45 days to reach the figure of five million.
- Comparing the pace of the vaccinations in the country with other countries, the additional secretary said that India is the fastest to inoculate more than 5 million beneficiaries against the deadly virus.
- In just 21 days, India vaccinated over 5.2 million beneficiaries, while the US took 24 days, the UK 43 days and Israel took 45 days to reach the figure of five million.
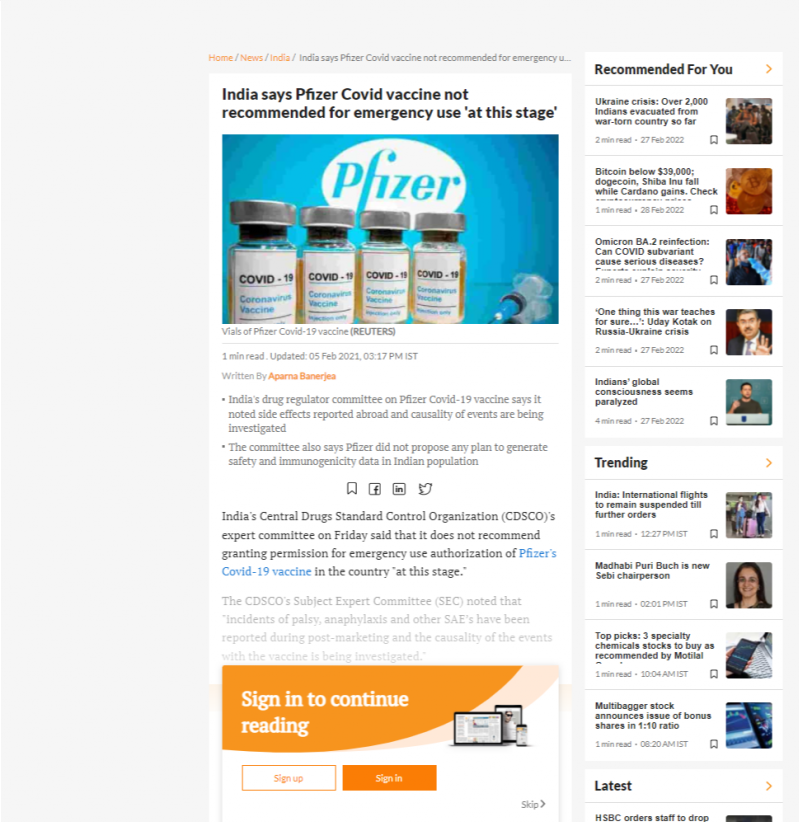
20487. India says Pfizer Covid vaccine not recommended for emergency use 'at this stage'
India Fights Corona: Vaccination Drive and Initiatives
English
India
Mint
|
- India's drug regulator committee on Pfizer Covid-19 vaccine says it noted side effects reported abroad and causality of events are being investigated
- The committee also says Pfizer did not propose any plan to generate safety and immunogenicity data in Indian population
- The CDSCO's Subject Expert Committee (SEC) noted that "incidents of palsy, anaphylaxis and other SAE’s have been reported during post-marketing and the causality of the events with the vaccine is being investigated."
20488. India: Pfizer withdraws COVID vaccine application for emergency use
India Fights Corona: Vaccination Drive and Initiatives
English
India
Dw.com
|
- World The company has said it would submit another authorization request with further information, as India said it had not seen enough evidence the shot was safe.
- Company failed to present evidence that its vaccine was safe for the Indian population, according to a Central Drugs Standard Control Organization (CDSCO) statement.
- India's drug regulator wanted Pfizer to conduct local trials to ensure the safety of the vaccine for official authorization. But the company failed to present evidence that its vaccine was safe for the Indian population, according to a Central Drugs Standard Control Organization (CDSCO) statement.
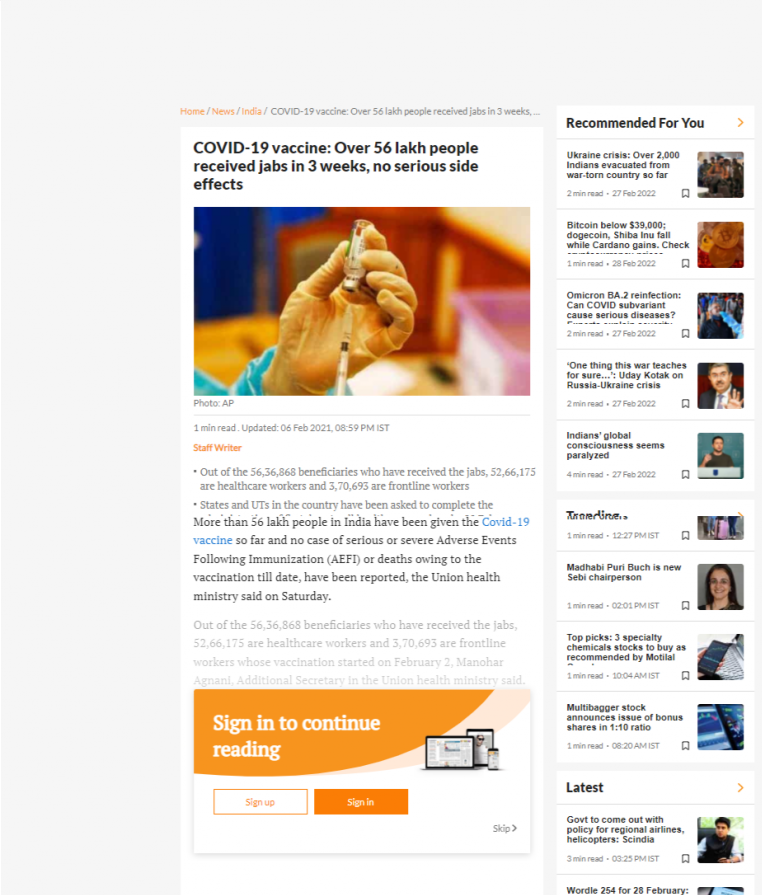
20489. COVID-19 vaccine: Over 56 lakh people received jabs in 3 weeks, no serious side effects
India Fights Corona: Vaccination Drive and Initiatives
English
India
Mint
|
- Out of the 56,36,868 beneficiaries who have received the jabs, 52,66,175 are healthcare workers and 3,70,693 are frontline workers
- States and UTs in the country have been asked to complete the administration of first dose to all health care workers by 20 February
- Out of the 56,36,868 beneficiaries who have received the jabs, 52,66,175 are healthcare workers and 3,70,693 are frontline workers whose vaccination started on February 2, Additional Secretary in the Union health ministry said.
20490. India developing 7 more COVID-19 vaccines: Health minister
Make in India / Atmanirbhar
->
Make in India- Public Health and Infrastructure
English
India
Mint
|
- Various Centre does not have any immediate plan to make the vaccines available in the open market, said health minister
- 'The COVID-19 inoculation process for people above 50 years of age will start next month,' he said
- Three of the vaccines are in the trial phase, two are in the pre-clinical stage, one is in phase 1 and another in phase 2, the Union health minister said.