Loading...
Loading...
| Jan 01, 1970 | Daily Report |
| RISING BHARAT | News Count (102633) | |
20381. Bharat Biotech, Serum Institute seek funds worth Rs 100 crore to ramp up Covid vaccine production
India Fights Corona: Vaccination Drive and Initiatives
English
India
India Today
|
- Bharat Biotech has written to the central government to seek funds worth Rs 100 crore to ramp up production of the coronavirus vaccine, 'Covaxin'.
- Sources also confirmed to India Today TV that the Serum Institute of India (SII) has also verbally requested for funds from the government’s Covid Suraksha scheme to increase their production
- Bharat Biotech is producing 4 million doses of Covaxin per month at their Hyderabad-based plant.
- Serum Institute had planned ramping up production to produce 100 million doses of Covishield by March 2021. The inter-ministerial group had also visited Serum Institute of India’s production manufacturing plant in Pune on March 21 regarding their ramping up of production.
- India is currently using two vaccines- Covishield which is manufactured by the Serum Institute of India, and Covaxin which is being manufactured by Bharat Biotech.
20382. भारत बायोटेक की वैक्सीन के क्लीनिकल ट्रायल में तीसरे डोज का भी होगा परीक्षण, मंजूरी मिली
India Fights Corona: Vaccination Drive and Initiatives
Hindi
India
ABP Live
|
- भारत बायोटेक की वैक्सीन के क्लीनिकल ट्रायल में तीसरा डोज भी दिया जाएगा. इस क्लीनिकल ट्रायल के लिए भारत बायोटेक को सेंट्रल ड्रग स्टैण्डर्ड कंट्रोलर आर्गेनाईजेशन की सब्जेक्ट एक्सपर्ट कमिटी से अनुमति मिल गई है.
- वैक्सीन के दूसरे चरण का ये एक्सटेंशन है. इसमे वैक्सीन की दूसरी डोज के 6 महीने बाद ये तीसरी डोज दी जाएगी. इसके बाद अगले 6 महीने तक इसका फॉलोअप किया जाएगा. इस ट्रायल में तीसरी डोज 6 माइक्रोग्राम की होगी.
- पहली और दूसरी डोज़ के बाद करीब 81% एफिकेसी मिलती है जो क्लीनिकल ट्रायल से सामने आया है. ऐसे में अगर तीसरी डोज दी जाए तो क्या एफिकेसी बढ़ेगी इस पर रिसर्च होगी.
20383. Australia to continue AstraZeneca vaccination despite blood clotting case
India Fights Corona: Vaccination Drive and Initiatives
English
Australia
The Times Of India
|
- MELBOURNE: Australia will continue its inoculation programme with AstraZeneca PLC, health officials said on Saturday, after a blood clotting case raised concern about the safety of the vaccine.
- The Therapeutic Goods Administration (TGA) regulator and a panel, the Australian Technical Advisory Group on Immunisation (ATAGI), met late Friday and early Saturday to discuss further advice on the AstraZeneca vaccine.
- On Thursday, Britain identified 30 cases of rare blood clot events following use of the vaccine. Several nations, including Canada, France, Germany and Spain, limited its use after similar reports.
- Possible complications with the rollout of the AstraZeneca vaccine could further slow the already delayed inoculation drive in Australia.
20384. 7 Recipients of AstraZeneca COVID-19 Vaccine Die in UK After Reporting Blood Clots
India Fights Corona: Vaccination Drive and Initiatives
English
United Kingdom
Deccanherald
|
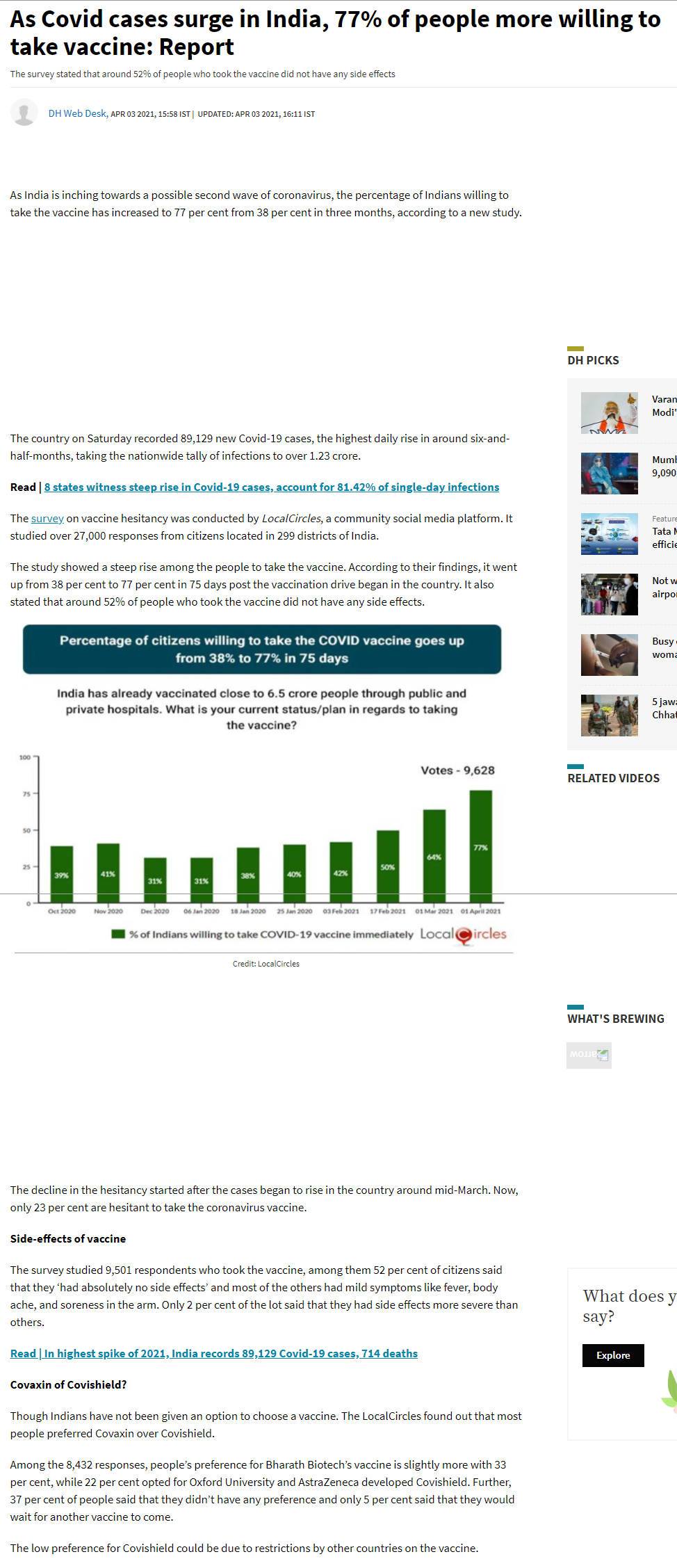
- India is inching towards a possible second wave of coronavirus, the percentage of Indians willing to take the vaccine has increased to 77 per cent from 38 per cent in three months, according to a new study.
- The survey stated that around 52% of people who took the vaccine did not have any side effects.
- The study showed a steep rise among the people to take the vaccine. According to their findings, it went up from 38 per cent to 77 per cent in 75 days post the vaccination drive began in the country. It also stated that around 52% of people who took.
20385. As Covid cases surge in India, 77% of people more willing to take vaccine: Report
India Fights Corona: Vaccination Drive and Initiatives
English
India
Deccanherald
|
- As India is inching towards a possible second wave of coronavirus, the percentage of Indians willing to take the vaccine has increased to 77 per cent from 38 per cent in three months, according to a new study.
- The survey stated that around 52% of people who took the vaccine did not have any side effects.
- The study showed a steep rise among the people to take the vaccine. According to their findings, it went up from 38 per cent to 77 per cent in 75 days post the vaccination drive began in the country. It also stated that around 52% of people who took ..
20386. Panacea Biotec to make 100 million doses of Sputnik V vaccine
India Fights Corona: Vaccination Drive and Initiatives
English
India
Mint
|
- Panacea Biotec Ltd on Monday announced that it had signed an agreement with Russian Direct Investment Fund (RDIF) for manufacturing 100 million doses of Sputnik V vaccine per year.
- The covid-19 vaccine is among those with the highest efficacy to be available in India.Panacea Biotec is the first experienced vaccine manufacturer in India with whom RDIF has a manufacturing agreement
- Production of Sputnik V at Panacea Biotec sites will help facilitate global supply of Sputnik V to international partners of RDIF, the two companies said in the statement.
20387. Serum Institute may seek permission for commercial sale of Covishield: Report
India Fights Corona: Vaccination Drive and Initiatives
English
India
Moneycontrol
|
- Serum Institute of India (SII) may apply for ‘marketing authorisation’ for Covishield, the AstraZeneca vaccine against COVID-19, which would allow the company to sell the vaccine commercially.
- Pune-based SII might apply for marketing authorisation of the jab to the Central Drugs Standard Control Organisation (CDSCO) by the end of April.
- SII is manufacturing and conducting clinical trials of the vaccine developed by British Swedish drugmaker AstraZeneca and the University of Oxford.
- The vaccine has been approved for restricted emergency use in India. Currently, no company in the world is allowed to sell vaccines against COVID-19 in the private market.
20388. 90% असरदार होगी कोविशील्ड वैक्सीन', अदार पूनावाला ने बताया दो डोज के बीच का सही अंतर
India Fights Corona: Vaccination Drive and Initiatives
Hindi
India
One India
|
- सीरम इंस्टीट्यूट ऑफ इंडिया को सीईओ अदार पूनावाला ने बताया है कि कोरोना वायरस के खिलाफ दी जा रही कोविशील्ड वैक्सीन अगर दो से तीन महीने के अंतर पर दी जाती है तो यह 90 प्रतिशत तक असरदार होगी।
- आक्सफोर्ड और एस्ट्राजेनेका के सहयोग से विकसित कोविशील्ड वैक्सीन का निर्माण भारतीय कंपनी सीरम इंस्टीट्यूट ऑफ इंडिया कर रही है।
- अदार पूनावाला ने इंडिया टीवी से बातचीत में बताया कि कोविशील्डि वैक्सीन का दो समूहों पर अध्ययन किया गया था।
- "एक समूह के लोगों को 1 महीने के अंतर पर वैक्सीन की दोनों डोज दी गई थी, उनमें 60-70 प्रतिशत असरदार रही। दूसरे समूह को 2 से 3 महीने के अंतर पर वैक्सीन दी गई। ऐसे लोगों में वैक्सीन 90 प्रतिशत प्रभावी पाई गई है।"
20389. Novavax to begin COVID vaccine trial on children, teenagers by second quarter
India Fights Corona: Vaccination Drive and Initiatives
English
India
CNBC TV 18
|
- American vaccine development company Novavax will soon kick off its pediatric trial for the COVID-19 vaccine.
- The officials of the company — whose COVID-19 vaccine jabs are also developed in the Serum Institute of India among other locations across the world — on Monday (April 5) said it will expand its vaccine trials to include children and teenagers by the second quarter.
- These trials are likely to be carried out in the United States (US) and Mexico, according to reports.
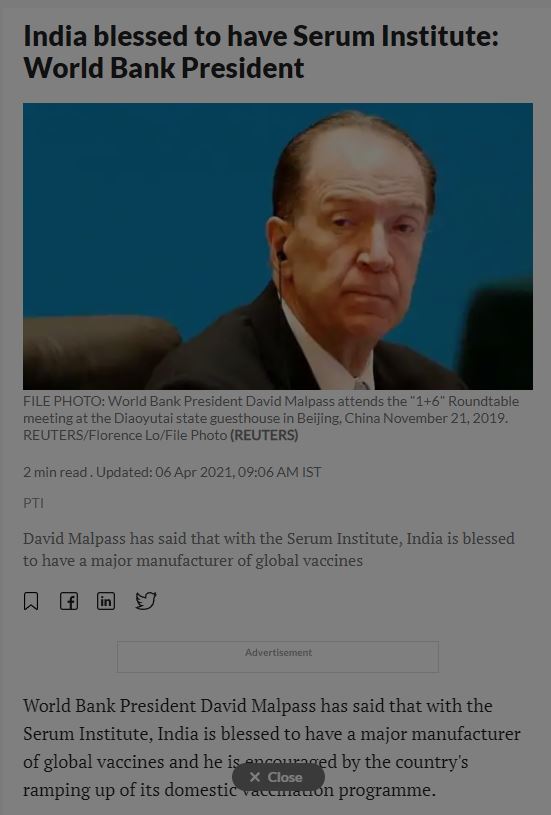
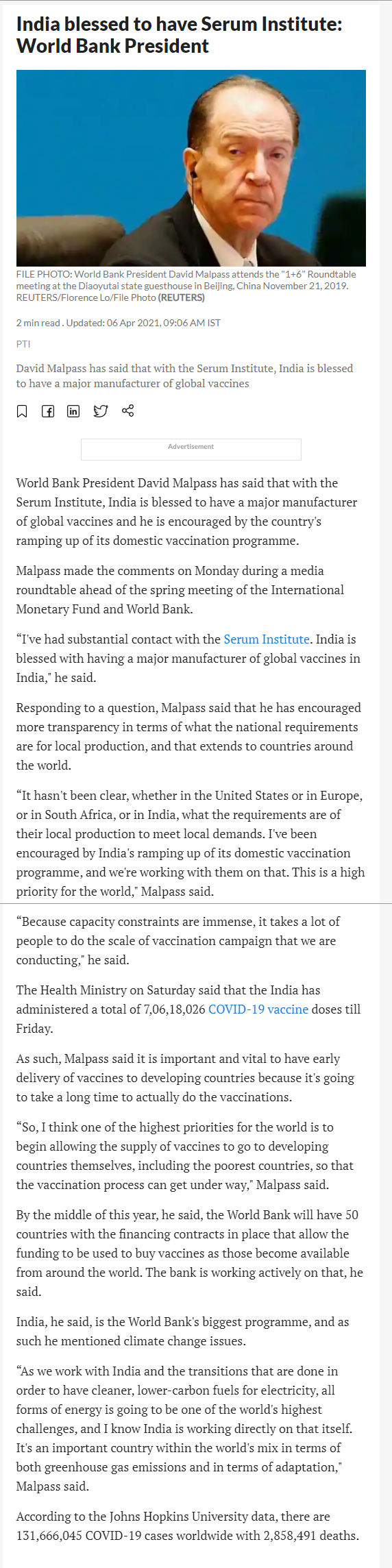
20390. India blessed to have Serum Institute: World Bank President
India Fights Corona: Vaccination Drive and Initiatives
English
India
Business Standard
|
- World Bank President David Malpass has said that with the Serum Institute, India is blessed to have a major manufacturer of global vaccines and he is encouraged by the country's ramping up of its domestic vaccination programme.
- David Malpass has said that with the Serum Institute, India is blessed to have a major manufacturer of global vaccines
- Malpass made the comments on Monday during a media roundtable ahead of the spring meeting of the International Monetary Fund and World Bank
- “I've had substantial contact with the Serum Institute. India is blessed with having a major manufacturer of global vaccines in India," he said.